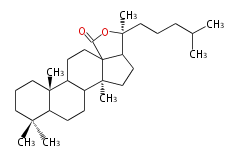Category:TP3
(→Design of Tri-terpene ID numbers ID番号の設計) |
|||
| Line 184: | Line 184: | ||
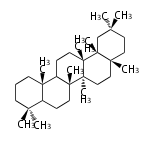
C30 (5 rings) | C30 (5 rings) | ||
hopane | hopane | ||
| + | FN | ||
| + | C30 (5 rings) | ||
| + | fernane | ||
CC | CC | ||
C30 | C30 | ||
| Line 205: | Line 208: | ||
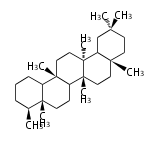
C30 (5 rings) | C30 (5 rings) | ||
oleanane | oleanane | ||
| + | FD | ||
| + | C30 (5 rings) | ||
| + | friedelane | ||
TR | TR | ||
C30 (5 rings) | C30 (5 rings) | ||
Revision as of 11:53, 5 August 2010
Contents |
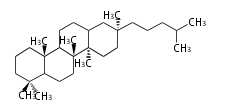
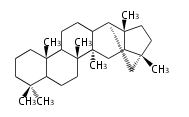
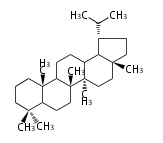
Triterpene (C30) Classes
Ring configuration
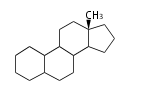
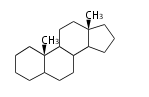
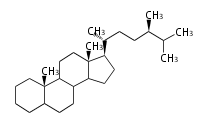
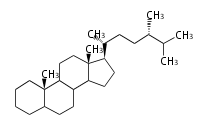
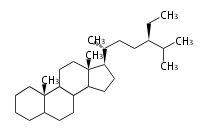
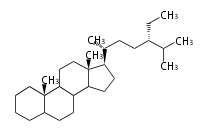
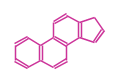
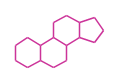
The basic structure is 4 carbon rings, cyclopenta[a]phenanthrene, gonane, or sterane. The rings B/C are always trans in all natural steroids. If the rings C/D are trans, it is called gonane. If its stereochemistry is unspecified, it is called sterane. Most steroids take gonane form, but in cardenolides and bufanolides, the rings C/D are cis.
 |
|
| Cyclopenta[a]phenanthrene | Gonane |
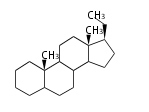
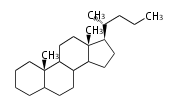
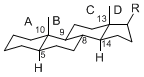
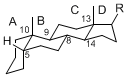
The majority of steroids have methyl groups sticking out from the bridgehead positions C-10 and C-13. When these methyl groups (or hydrogens) stand above the plane, they are called β-configuration. Those below the plane are called α-configuration. If the configuration at any site is unknown, it is indicated as ξ (Greek Xi). By default, hydrogen atoms or substituents at the positions C-8, 9, 10, 13, and 14 are assumed to be 8β, 9α, 10β, 13β, and 14α configurations. C-5 is a special position, because there are as many 5α steroids as 5β are.
 |
 |
|
| cholestane backbone | 5α-configuration | 5β-configuration |
Biosynthesis
The starting point is 2,3-oxidosqualene, which is formed by joining two FPPs tail-to-tail. This molecule undergoes cyclization with Wagner-Meerwein shift to form taxa-dependent precursors.
- animals, fungi, and yeast
- 2,3-oxidosqualene → lanosterol
- plants (including algae)
- 2,3-oxidosqualene → cycloartenol, dammarane
- bacteria
- 2,3-oxidosqualene → hopene
Design of Tri-terpene ID numbers ID番号の設計
12-DIGIT
| T | P | 3 | x | y | y | r | h | g | n | c | c |
- x ... species information
| Symbol at x | Kingdom | Phyla | Examples |
|---|---|---|---|
| I | Animalia | Arthropoda (Insects, crabs) | ecdysteroids |
| V | Chordate (Vertebrates) | sex steroids, corticosteroids, anabolic steroids | |
| O | Others | marine steroids | |
| P | Plantae | Phytosterols | lanosterols, cholesterols, brassinolides |
| S | Saponins | saponins | |
| F | Fungi | ergosterols | ergosterols |
| B | Bacteria | bacterial sterols | hopanoids |
- y ... backbone structure (母核構造)
- r ... number of major rings (環構造数)
Click above categories to see details.
- h ... hydroxylation pattern (水酸基数)
Click above categories to see details.
- g ... glycosylation pattern(糖修飾パターン)
Click above categories to see details.
- n ... number of sugars (修飾糖数)
Click above categories to see details.
- c ... serial number (通し番号)
Cite error:
<ref> tags exist, but no <references/> tag was found
This category currently contains no pages or media.