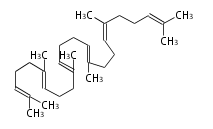
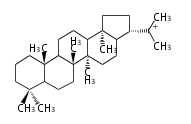
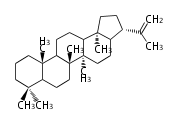
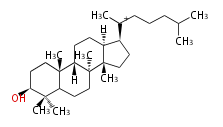
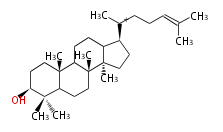
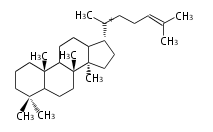
Triterpene (C30) Classes
Ring configuration
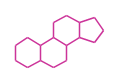
The basic structure is 4 carbon rings, cyclopenta[a]phenanthrene, gonane, or sterane.
The rings B/C are always trans in all natural steroids. If the rings C/D are trans, it is called gonane. If its stereochemistry is unspecified, it is called sterane.
Most steroids take gonane form, but in cardenolides and bufanolides, the rings C/D are cis.
 |
|
| Cyclopenta[a]phenanthrene
|
Gonane
|
The majority of steroids have methyl groups sticking out from the bridgehead positions C-10 and C-13.
When these methyl groups (or hydrogens) stand above the plane, they are called β-configuration.
Those below the plane are called α-configuration. If the configuration at any site is unknown, it is indicated as ξ (Greek Xi).
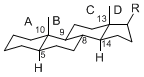
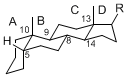
By default, hydrogen atoms or substituents at the positions C-8, 9, 10, 13, and 14 are assumed to be
8β, 9α, 10β, 13β, and 14α configurations. C-5 is a special position, because there are as many 5α steroids as 5β are.
 |
 |
|
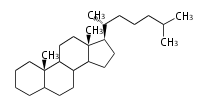
| cholestane backbone
|
5α-configuration
|
5β-configuration
|
Biosynthesis
The starting point is squalene, which is formed by joining two FPPs tail-to-tail.
Bacterial cyclases use squalene directly, but those of the other species use
2,3-oxidosqualene for cyclization.
- In bacteria, squalene is cyclized via the 17α-deoxydammarenyl cation to hopene and other triterpenes[1].
- In eukaryotes, 2,3-oxidosqualene is cyclized via the protosteryl cation to lanosterol (animals and fungi) or cycloartenol (plants) by a series of 1,2-hydride and methyl shifts (Wagner-Meerwein shifts).[2]
- In plants, various triterpenes arise from the dammarenyl cation.
- ↑ Bacterial squalene cyclases can accept oxidosqualene as their substrates, but oxidosqualene usually does not exist in bacteria
- ↑ A trace amount of phytosterols comes from lanosterol. Ohyama K, Suzuki M, Kikuchi J, Saito K, Muranaka T (2009) Dual biosynthetic pathways to phytosterol via cycloartenol and lanosterol in Arabidopsis Proc Natl Acad Sci USA 106(3):725-730
- ↑ The most accessible enzyme among oxidosqualene cyclases.
Ref. Corey EJ, Russey WE, Ortiz-de-Montellano PR (1966) 2,3-Oxidosqualene, an intermediate in the biological synthesis of sterols from squalene J Am Chem Soc 88:4750-1
- ↑ The rings of protosteryl cation are chair-boat-chair configuration. The C-17 chain is β-configuration, not α.
Ref. Corey EJ, Virgil SC (1991) An experimental demonstration of the stereochemistry of enzymic cyclization of 2,3-oxidosqualene to the protosterol system, forerunner of lanosterol and cholesterol. J Am Chem Soc 113:4025-6
- ↑ The rings of dammarenyl cation are all-chair configuration. The C-17 chain is β-configuration.
Ref. Xiong Q, Rocco F, Wilson WK, Xu R, Ceruti M, Matsuda SPT (2005) Structure and reactivity of the dammarenyl cation: configuration transmission in triterpene synthesis. J Org. Chem. 70:5362-75
Useful Reviews:
- Xu R, Fazio GC, Matsuda SPT (2004) On the origins of triterpenoid skeletal diversity. Phytochemstry 65:261-291 PMID 14751299
Design of Tri-terpene ID numbers ID番号の設計
12-DIGIT
- x ... species information
-
- y ... backbone structure (母核構造)
-
| Symbol at y |
Carbons |
Steroids |
Backbone Structure
|
| GN
|
C17
|
gonane
|

|
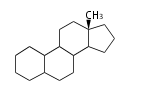
| ES
|
C18
|
estrane
|
|
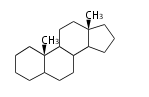
| AD
|
C19
|
androstane
|
|
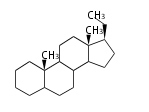
| PG
|
C21
|
pregnane
|
|
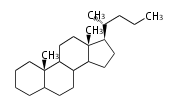
| CA
|
C24
|
cholane
|
|
| CL
|
C27
|
cholestane
|

|
| CM
|
C28
|
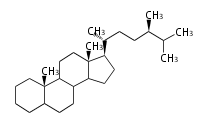
campestane
|
|
| EG
|
C28
|
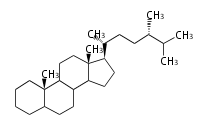
ergostane
|
|
| SG
|
C29 (4 rings)
|
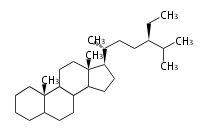
stigmastane
|
|
| PR
|
C29 (4 rings)
|
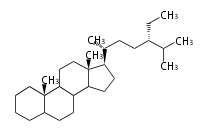
poriferastane
|
|
| Symbol at y |
Carbons |
Triterpenoids |
Backbone Structure
|
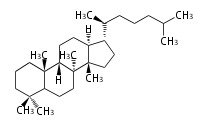
| PS
|
C30
|
protostane
|
|
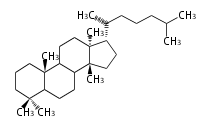
| EU
|
C30
|
euphane
|
|
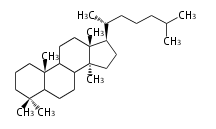
| LN
|
C30
|
lanostane
|
|
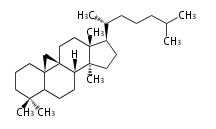
| CY
|
C30
|
cycloartane
|
|
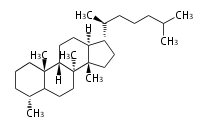
| FS
|
C29
|
fusidane
|
|
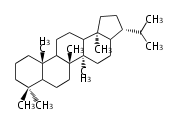
| HP
|
C30 (5 rings)
|
hopane
|
|
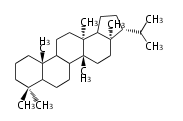
| FN
|
C30 (5 rings)
|
fernane
|
|
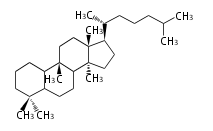
| CC
|
C30
|
cucurbitane
|
|
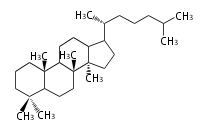
| DM
|
C30
|
dammarane
|
|
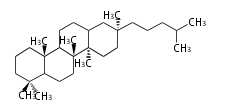
| BC
|
C30
|
baccharane
|
|
| HL
|
C30
|
holostane
|
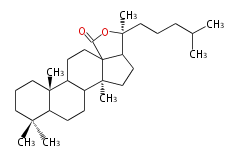
|
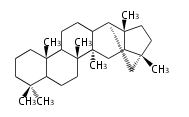
| PF
|
C29 (5 rings)
|
pfaffane
|
|
| LP
|
C30 (5 rings)
|
lupane
|
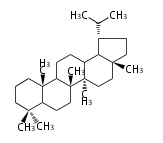
|
| OL
|
C30 (5 rings)
|
oleanane
|
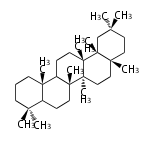
|
| FD
|
C30 (5 rings)
|
friedelane
|
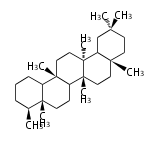
|
| TR
|
C30 (5 rings)
|
taraxastane
|
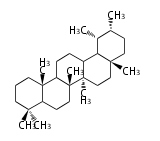
|
| UR
|
C30 (5 rings)
|
ursane
|
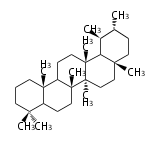
|
| SR
|
C30 (5 rings)
|
serratane
|
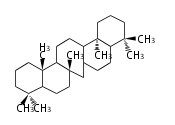
|
- r ... number of major rings (環構造数)
-
Click above categories to see details.
- h ... hydroxylation pattern (水酸基数)
-
Click above categories to see details.
- g ... glycosylation pattern(糖修飾パターン)
-
Click above categories to see details.
- n ... number of sugars (修飾糖数)
-
Click above categories to see details.
- c ... serial number (通し番号)
Cite error: <ref> tags exist, but no <references/> tag was found
This category currently contains no pages or media.