|
|
| Line 6: |
Line 6: |
| | }} | | }} |
| | | | |
| − | ==Steroid==
| |
| − | ===Ring configuration===
| |
| − | {{Twocolumn|
| |
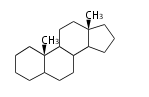
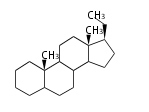
| − | The basic steroid structure is 4 carbon rings, cyclopenta[a]phenanthrene, gonane, or sterane.
| |
| − | The rings B/C are always ''trans'' in all natural steroids. If the rings C/D are ''trans'', it is called gonane. If its stereochemistry is unspecified, it is called sterane.
| |
| − | Most steroids take gonane form, but in cardenolides and bufanolides, the rings C/D are ''cis''.
| |
| − | |
| |
| − | ステロイドの基本骨格は4つの環状構造で、シクロペンタ[a]フェナンスレン、ゴナン、ステランなどと呼ばれる。
| |
| − | 天然のステロイドでは環 B/C は常にトランスの位置にある。環 C/D がトランスの場合をゴナンと呼び、立体配置が指定されていないときをステランと呼ぶ。
| |
| − | ほとんどのステロイドはゴナンの形をとるが、カルデノライドとブファノライドは環 C/D がシスになる。
| |
| − | }}
| |
| − |
| |
| − | <center>
| |
| − | {| style="text-align:center"
| |
| − | | [[Image:Cyclopenta-a-phenanthrene.png]] || [[Image:Gonane.png]]
| |
| − | |-
| |
| − | | Cyclopenta[a]phenanthrene
| |
| − | | Gonane
| |
| − | |}
| |
| − | </center>
| |
| − |
| |
| − | {{Twocolumn|
| |
| − | The majority of steroids have methyl groups sticking out from the bridgehead positions C-10 and C-13.
| |
| − | When these methyl groups (or hydrogens) stand above the plane, they are called β-configuration.
| |
| − | Those below the plane are called α-configuration. If the configuration at any site is unknown, it is indicated as ξ (Greek Xi).
| |
| − | By default, hydrogen atoms or substituents at the positions C-8, 9, 10, 13, and 14 are assumed to be
| |
| − | 8β, 9α, 10β, 13β, and 14α configurations. C-5 is a special position, because there are as many 5α steroids as 5β are.
| |
| − | |
| |
| − | 大多数のステロイドは橋頭位のC-10, C-13からメチル基が出ている。
| |
| − | これらのメチル基 (または水素など) が平面より上に出ているときを、ベータ配置とする。平面より下の場合をアルファ配置とする。
| |
| − | また、配置が不明な場合は、ξ (ギリシャ語の Xi)で表す。
| |
| − | 原則として、C-8, 9, 10, 13, 14位にある水素や置換基はそれぞれ8β, 9α, 10β, 13β, 14α 配置である。
| |
| − | C-5位は特別であり、5α のステロイドと 5β のものが同じくらい存在する。
| |
| − | }}
| |
| − |
| |
| − | <center>
| |
| − | {| style="text-align:center"
| |
| − | | [[Image:5alpha-steroid.png]] || [[Image:5beta-steroid.png]]
| |
| − | |-
| |
| − | | 5α-configuration
| |
| − | | 5β-configuration
| |
| − | |}
| |
| − | </center>
| |
| − |
| |
| − | ===Hormonal Classes===
| |
| − | ====Estranes (C18)====
| |
| − | {{Twocolumn|
| |
| − | The backbone of estrogens, the hormones responsible for female reproductive organs and secondary sex characteristics. The original spelling was 'oestrogen'. Major namings are:
| |
| − | * estrogen ... estra-1,3,5(10)-triene
| |
| − | * estrone ... 3-hydroxyestra-1,3,5(10)-triene-17-one
| |
| − | * estradiol ... estra-1,3,5(10)-triene-3,17β-diol
| |
| − | * estriol ... estra-1,3,5(10)-triene-3,16α,17β-triol
| |
| − | * equine estrogens ... estra-1,3,5(10),7-tetraene and estra-1,3,5,7,9-pentaene series
| |
| − | |
| |
| − |
| |
| − | }}
| |
| − |
| |
| − | ====Androstanes (C19)====
| |
| − | {{Twocolumn|
| |
| − | The backbone of male hormone testosterones. Major namings are:
| |
| − | * etianic acids (C20) ... androstane-17-carboxylic acids and their derivatives
| |
| − | * D-homoandrostanes (C20) ... ring D is expanded by including C-17a carbon.
| |
| − | |
| |
| − |
| |
| − | }}
| |
| − |
| |
| − | ====Pregnanes (C21)====
| |
| − | {{Twocolumn|
| |
| − | The backbone of pregnancy hormone progesterone and the majority of corticosteroids.
| |
| − | It is the largest single group of steroids.
| |
| − | Major namings are:
| |
| − | * progesterone ... pregn-4-ene-3,20-dione
| |
| − | * 17α-ethynylandrostane ... 17α-pregn-20-yne
| |
| − | * 19-norpregnane (C20) ... 17α-ethynylestrane, 19-nor-17α-pregn-20-yne
| |
| − |
| |
| − | 17α-pregnanes are sometimes called 17α-vinyl- and 17α-ethyl-androstanes.
| |
| − | |
| |
| − | }}
| |
| − |
| |
| − | ====Cholanes (C24)====
| |
| − | {{Twocolumn|
| |
| − | The backbone of bile acids, i.e., 5β-cholan-24-oic acids with hydroxyl substitution at C-3.
| |
| − | * primary bile acids ... cholic acid and chenodeoxycholic acid are biosynthesized directly from cholesterol.
| |
| − | * secondary bile acids ... deoxycholic acid and lithocholic acid are generated by intestinal bacteria.
| |
| − | * 24-norcholan-23-oic acids ... loss of one carbon side chain
| |
| − | * 23,24-dinorcholan-22-oic acids ... loss of two carbon side chains
| |
| − | |
| |
| − |
| |
| − | }}
| |
| − |
| |
| − | ===C/D ''cis'' Classes===
| |
| − |
| |
| − | ====Cardenolides (C23)====
| |
| − | {{Twocolumn|
| |
| − | The backbone of ''Digitalis'' glycosides with the androstane skeleton with a γ-lactone ring at C-17. Notable characters are its 14β-configuration in opposition to other steroids (the rings C/D are ''cis''), and the 20(22)-double bond. Their cardiac activity is well known.
| |
| − | |
| |
| − |
| |
| − | }}
| |
| − |
| |
| − | ====Bufanolides (C24)====
| |
| − | {{Twocolumn|
| |
| − | The backbone of toad skin secretions and the sea onion or squill (''Scilla maritima'') with a δ-lactone ring at C-17.
| |
| − | As for cardenolides, 14β, 17β, and 20''R''-configurations are assumed (the rings C/D are ''cis''). They naturally occur as glycosides or conjugates of suberylarginine.
| |
| − | |
| |
| − | }}
| |
| − |
| |
| − | ===Steroidal Classes===
| |
| − |
| |
| − | ====Sterols (C27)====
| |
| − | {{Twocolumn|
| |
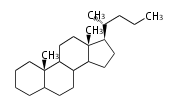
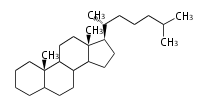
| − | The sterol backbone contains a β hydroxyl group at C-3 and side chains of 8-10 carbons at C-17. They are important membrane constituents in addition to their biologically activity.
| |
| − | <br/>
| |
| − | Major categories are:
| |
| − | * cholestane ... androstane structure with a branched C8 chain at the 17β-position
| |
| − | * norcholestane ... cholestane lacking one of 27 carbons
| |
| − | * ergostane and campestane ... 24β- and 24α-methylcholestane
| |
| − | * poriferastane and stigmastane ... 24β- and 24α-ethylcholestane
| |
| − | * vitamin D2 ... 9,10-secoergonstane derivatives
| |
| − | * vitamin D3 ... 9,10-secocholestane derivatives
| |
| − | * lumistane ... 9β,10α-ergosta-5,7-diene
| |
| − | |
| |
| − | }}
| |
| − |
| |
| − | ====Ecdysteroid (C27)====
| |
| − | {{Twocolumn|
| |
| − | Ecdysteroids (or ecdysones) are moulting hormones of insects and crustaceans, but also present in many plants.
| |
| − | The basic skeleton is highly oxygenated cholestane: 2β,3β,14α,20,22-pentahydroxy-5β-cholest-7-en-6-one.
| |
| − | The first ecdysteroid to be isolated was α-ecdysone from teh silkworm (''Bombyx mori'').
| |
| − | |
| |
| − | }}
| |
| − |
| |
| − | ====Spirostans and furostans (C27)====
| |
| − | {{Twocolumn|
| |
| − | Furostan has the additional epoxy ring E. Spirostan has another epoxy ring F, which is perpendicular to the planar orientation of rings A-E. The omission of a terminal 'e' from spirostans and furostans indicates that they are not hydrocarbons.
| |
| − | (However, the last 'e' is needed if a consonant follows.)
| |
| − | <br/>
| |
| − | Major categories are:
| |
| − | * furostan ... 16β,22-epoxycholestane
| |
| − | * spirostan ... 22,26-epoxyfurostan
| |
| − | |
| |
| − |
| |
| − | }}
| |
| − |
| |
| − | ====Withanolides (C28)====
| |
| − | {{Twocolumn|
| |
| − | Withanolides were found in the root of [[Species:Withania|Withania Somnifera]], also known as Indian ginseng. Its backbone is a highly oxygenated ergostane with a γ-lactone ring linking C-22 and C-26 <ref>Iida, T et al. (1985) J. Lipid Res. 26,874</ref>. The configuration of C-22 is usually ''R''.
| |
| − | |
| |
| − | }}
| |
| − |
| |
| − | ====Brassinolides (C28)====
| |
| − | {{Twocolumn|
| |
| − | Brassinolides are plant growth-promoting hormones isolated originally from [[Species:Brassica|''Brassica napus'']] (rape). Its backbone is a highly oxygenated ergostane with the oxygen-expanded B-ring (ε-lactone). This lactone is not essential for plant growth activity (e.g. castasterone) but the 22''R'', 23''R''-diol are.
| |
| − | The configurations of C-2,3 and 5 are α in brassinolides whereas they are β in ecdysteroids.
| |
| − | |
| |
| − |
| |
| − | }}
| |
| − |
| |
| − | ====Gorgostane (C30)====
| |
| − | {{Twocolumn|
| |
| − | Gorgostanes occur in marine organisms. Its backbone is ergostane with an additional metyl group at C-23 and ana methylene bridge between C-22 and C-23.
| |
| − | |
| |
| − |
| |
| − | }}
| |
| − |
| |
| − | ===Biosynthesis===
| |
| − | {| style="text-align:center; cellpadding:0"
| |
| − | |-
| |
| − | | [[Image:2,3-oxidosqualene.png]]<br/>2,3-Oxidosqualene
| |
| − | | [[Image:Arrow00r.png]]
| |
| − | | [[Image:Dammarane.png]]<br/>{{Bilingual|Dammarane-type|ダンマラン (dammarane) 型}}
| |
| − | |-
| |
| − | | || || [[Image:Arrow00d35.png]]
| |
| − | |-
| |
| − | | [[Image:Cholestane.png]]<br/>Cholestane-type
| |
| − | | [[Image:Arrow00l.png]]
| |
| − | | [[Image:Lanostane.png]]<br/>Lanostane-type
| |
| − | |-
| |
| − | | [[Image:Arrow00d35.png]]
| |
| − | |-
| |
| − | | [[Image:Pregnane.png]]<br/>Pregnane-type
| |
| − | | [[Image:Arrow00r.png]]
| |
| − | | [[Image:Androstane.png]][[Image:Estrane.png]]<br/>Androstane- and Estrane-type
| |
| − | |}
| |
| | | | |
| | ==Phytosterols== | | ==Phytosterols== |
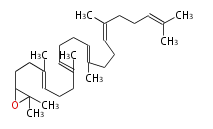
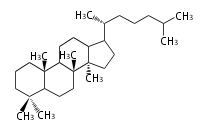
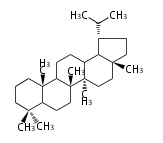
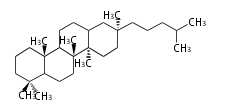
Triterpenes are formed by joining two FPPs tail-to-tail. The precursor compound of cholesterol (C27) is lanosterol (C30) for animals. For plants, fungi and algae, it is almost cycloartenol with a trace of lanosterol-derived sterols[1].
Most common phytosterols are campesterol, β-sitosterol, and stigmasterol. Soybean (Glycine max, Fabaceae) is a rich source of phytosterols (about 0.1% of its weight), and is used for semi-synthesis of medicinal steroids [2]. Since dietary phytosterols reduce cholesterol levels, they are used as food additives such as for margarine [3]. Vitamin D is a family of sterol metabolites generated photochemically in our skin by UV irradiation.
Saponins are surfactant glycosides, i.e., they produce foams in aqueous solution and therefore are called ‘sapo’ (a Latin for soap). Plant-based crude drugs are still actively prescribed in Eastern Asia, and many of their active components are attributed to saponins. Well known examples of saponin and its aglycone include glycyrrhizin from liquorice (Glycyrrhiza uralensis/glabra, Category:Fabaceae) used in European confectionery and Asian medicine; ginsenosides from ginseng (Panax ginseng, Araliaceae) for tonic, especially in Korea; diosgenin from wild yam (Dioscorea spp., Dioscoreaceae) for hormone replacement therapy.
Click above categories to see details.
Click above categories to see details.
Click above categories to see details.
Click above categories to see details.