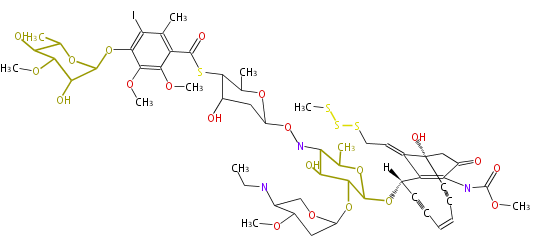
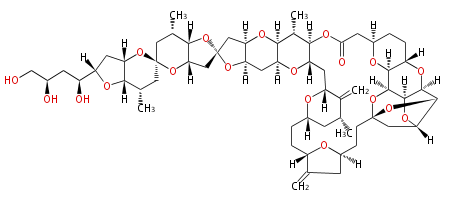
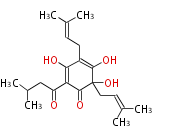
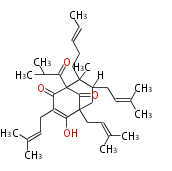
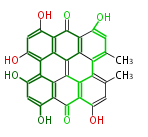
Polyketide
Class Overview
Polyketides are synthesized through the polymerization of acetyl units (β-ketomethylene) as in fatty acid biosynthesis. Typical starter units are short-chain fatty acids (e.g. acetyl-CoA or propionyl-CoA), on to which extender units (e.g. malonyl-CoA or methylmalonyl-CoA) are repeatedly polymerized.
The key reactions for the chain extension are:
- Claisen condensation by β-ketoacyl synthase (KS)
- an acyltransferase (AT), and
- an acyl carrier protein (ACP).
After elongation, β-ketone is reduced. In fatty acid biosynthesis, the chain is fully reduced by the following three steps:
- Reduction to an alcohol by ketoreductase (KR),
- Dehydration to the conjugated ester by dehydratase (DH), and
- Reduction of the double bond by enoyl reductase (ER).
In polyketide synthase, the reduction is patial.
Finally, the chain is terminated by a thioesterase (TE) activity and
allows Claisen cyclization (CYC).
Classification
3-4th digits
| Linear Chain and Related (L)
|
|
|
| Polyether (LE)
|
| nigericin
|
| monensin
|
| okadaic acid
|
| ciguatoxin, brevetoxin
|
| halichondrin
|
| zaragozic acid
|
|
Acetogenins (LA)
|
| Aromatic and Diels-Alder Related (most often by iterative type II)
|
| Monocyclic (A1)
|
| Salicylic acid
|
| orsellinic acid
|
| benzophenone
|
| altenariol
|
|
| Bicyclic (A2)
|
| lovastatin
|
| aflatoxins[1]
|
|
| Tricyclic (A3)
|
| emodin
|
| sennoside
|
| hypericin
|
|
| Tetracyclic (A4)
|
| Linear type
|
| Angucycline
|
|
|
| Macrolides (most often by non-iterative type I)
|
|
|
| Size14-ring (M4)
|
| Colletodiol
|
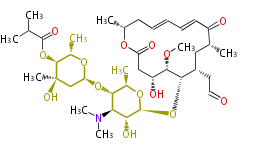
| Erythromycin[2]
|
| Fluvirucin
|
| Zearalenone
|
|
| Size16-ring (M6)
|
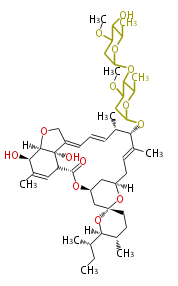
| Avermectin
|
| Bafilomycin
|
| Oligomycin
|
| Tylosin
|
|
| Polyene (MN)
|
| Manumycin
|
| Nystatin
|
| Amphotericin
|
|
| Polyether (ME)
|
| Nonactin, Nactin
|
|
| Cyclic Imines (MI)
|
| Spirolide
|
| Pinnatoxin
|
|
| Ansamacrolide (MA)
|
| Rifamycin
|
| Ansamycin
|
|
| Others (MO)
|
| Bryostatin (26)Cite error: Closing </ref> missing for <ref> tag
|
| Milbemycin (20)
|
| Myxovirescin (28)[3]
|
| Natamycin (26)[4]
|
| Tacrolimus (23)
|
|
- References
- ↑ foo
- ↑ 6-deoxy sugars (L-cladinose and D-desosamine) are attached.
- ↑ http://www.indiana.edu/~drwchem/pdfs/50.pdf
- ↑ =Pimaricin
5th digit
| The number of C2 unit
|
4 Units
orsellinic acid, 6-methylsalicylic acid, triacetic acid lactone, asperlin, usnic acid, methylphloracetophenone, penicillic acid, patulin
|
5 Units
citrinin, aflatoxin, augenone, sepedonin, stipitatonic acid
|
6 Units
plumbagin, 7-methyljuglone, juglone, variotin
|
7 & 8 Units
Anthraquinone rings
griseofulvin, rubrofusarin, emodin, alizarin, pachybasin, xanthone, versicolorin A, aflatoxin B1, sterigmatocystin, tajixanthone
|
9 Units
Tetracyclines
terramycin, aureomycin, daunomycin
|
>9 Units
|
Polyketide Synthase (PKS)
Distribution
PKS members are found in bacteria, fungi, plants, slime mold[1], Alveolata[2], and animals [3][4].
- References
- ↑ Zucko J, Skunca N, Curk T, Zupan B, Long PF et al (2007) "Polyketide synthase genes and the natural products potential of Dictyostelium discoideum" Bioinformatics 23:2543-49
- ↑ Zhu G, LaGier MJ, Stejskal F, Millership JJ, Cai X et al (2002) "Cryptosporidium parvum: the first protist known to encode a putative polyketide synthase" Gene 298:79-89
- ↑ Castoe TA, Stephens T, Noonan BP, Calestani C (2007) "A novel group of type I polyketide synthases (PKS) in animals and the complex phylogenomics of PKSs" Gene 392:47-58
- ↑ Calestani C, Rast JP, Davidson EH (2003) "Isolation of pigment cell specific genes in the sea urchin embryo by differential macroarray screening" Development 130:4587-96
Type I, II, and III
There are three types of PKSs known to date.
Type I : multiple domains per protein (e.g. Erythromycin biosynthesis [1])
- Bacterial type I is modular.
- Fungal type I is "iterative", i.e., it reuses same active sites through multiple catalytic steps. Non-reducing (NR) type produces aromatic polyketides, and partially reducing type produces others.
Type II : single domain per protein
- Three proteins (KSα, KSβ, ACP) are repeatedly used for carbon chain elongation, and the chain length is determined by another protein, CLF.
- In bacteria, products are aromatic (e.g. chiorotetracycline, pradimicin).
Type III : chalcone synthase-like in plants
- Discovered in plants, but later found in bacteria[2]
| Species |
Actinomycetes |
Cyanobacteria |
γ-Proteobacteria |
Fungi |
Alveolata
|
| Type-I PKS
|
Ο |
Ο |
Ο |
Ο |
Ο
|
| Type-II PKS
|
Ο |
Χ |
Χ |
Χ |
Χ
|
| NRPS
|
Ο |
Ο |
Ο |
Ο |
Χ
|
| deoxysugar
|
Ο |
Χ |
Χ |
Χ |
Χ
|
| Terpene
|
Δ |
Χ |
Χ |
Ο |
Χ
|
Non-ribosomal peptide synthase (NRPS)
Coupling with PKS and NRPS
- vancomycin ()
- leinamycin (Curr opin chem biol 7:285, 2003)
- pseurotin (chem bio chem 8:1736-1743, 2007)
- curacin (curr opin chem biol 13:216, 2009)
- epothilone
- rapamycin
Decoration
deoxysugars
deoxygenation, c-methylation, amination, n-methylation, ketosugar,
Unusual structures
| Phoma
|
zaragozic acid, phomoidoride
|
Streptomyces
|
yatakemycin, leinamycin, saframycin, neocarzinostatin, staurosporin, FR182877
|
Other bacteria
|
PKS-NRPS hybrid type
Curacin A (Lyngbya), Shiphonazole (Herpetosiphon), Jamaicamide A (Lyngbya), Cylindrospermopsin (Cylindrospermopsis)
|
Unusual PKSs
- Bacterial but iterative type I PKS for aromatic polyketide
- AviM for orsellinic acid biosynthesis (Streptomyces viridochromogens Tu57)[3]
- CalO5 for calicheamicin biosyntehsis (Micromonospora echinospora ssp. calichenisis)[4]
- NesB for neocarzinostatin biosynthesis (?)[5]
- Type I PKS that lacks teh cognate AT domain
- lnmIJ for leinamycin biosynthesis (Streptomyces atroolivaceus S-140)[6]
- PedF for pederin biosynthesis (symbiont bacterium of Paederus beetles)[7]
- Type II PKS that act non-iteratively and use acyl CoA as substrates directly
- NonPQU and NonJK (Streptomyces griseus)[8]
- ↑ (2001) Nat Prod Rep 18:380
- ↑ Moore BS, Hopke JN (2001) Discovery of a new bacterial polyketide biosynthetic pathway Chembiochem 2:35-8
- ↑ Gaisser S, Trefzer A, Stockert S, Kirschning A, Bechthold A (1997) Cloning of an avilamycin biosynthetic gene cluster from Streptomyces viridochromogenes Tu57. J Bacteriol 179:6271-8
- ↑ Whitwam RE, Ahlert J, Holman TR, Ruppen M, Thorson JS (2000) The gene calC encodes for a non-heme iron metalloprotein responsible for calicheamicin self-resistance in Micromonospora. J Am Chem Soc 122:1556-7
- ↑ Zazopoulos E, Huang K, Staffa A, Liu W, Bachmann BO, Nonaka K, Ahlert J, Thorson JS, Shen B, Farnet CM (2003) A genomics-guided approach for discovering and expressing cryptic metabolicpathways Nat Biotechnol epub.
- ↑ Cheng Y-Q, Tang G-L, Shen B (2003) Type I polyketide synthase requiring a discrete acyltransferase for polyketide biosynthesis Proc Natl Acad Sci U S A 100: in press
- ↑ Piel J (2002) A polyketide synthase-peptide synthetase gene cluster from an uncultured bacterial symbiont of Paederus beetles Proc Natl Acad Sci U S A 98:14808-13
- ↑ Kwon HJ, Smith WC, Scharon AJ, Hwang SH, Kurth MJ, Shen B (2002) C-O bond formation by polyketide synthases Science 297(5585):1327-30
Subcategories
This category has only the following subcategory.