FL3FECGS0041
From Metabolomics.JP
(Difference between revisions)
| (3 intermediate revisions by one user not shown) | |||
| Line 1: | Line 1: | ||
| + | {{Hierarchy|{{PAGENAME}}}} | ||
| + | |||
{{Metabolite | {{Metabolite | ||
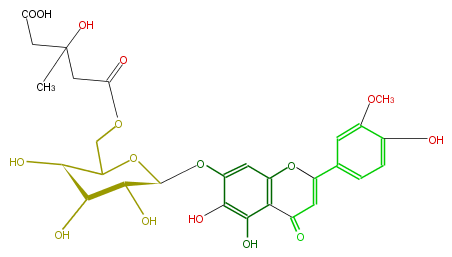
| − | |SysName=7-[[6-O-(4-Carboxy-3-hydroxy-3-methyl-1-oxobutyl)-beta-D-glucopyranosyl]oxy]-5,6-dihydroxy-2-(4-hydroxy-3-methoxyphenyl)-4H-1-benzopyran-4-one | + | |SysName=7- [ [ 6-O- (4-Carboxy-3-hydroxy-3-methyl-1-oxobutyl) -beta-D-glucopyranosyl ] oxy ] -5,6-dihydroxy-2- (4-hydroxy-3-methoxyphenyl) -4H-1-benzopyran-4-one |
| − | |Common Name=&&6-Hydroxyluteolin 3'-methyl ether 7-[6"-(3-hydroxy-3-methylglutaryl)glucoside]&&7-[[6-O-(4-Carboxy-3-hydroxy-3-methyl-1-oxobutyl)-beta-D-glucopyranosyl]oxy]-5,6-dihydroxy-2-(4-hydroxy-3-methoxyphenyl)-4H-1-benzopyran-4-one&& | + | |Common Name=&&6-Hydroxyluteolin 3'-methyl ether 7- [ 6"- (3-hydroxy-3-methylglutaryl) glucoside ] &&7- [ [ 6-O- (4-Carboxy-3-hydroxy-3-methyl-1-oxobutyl) -beta-D-glucopyranosyl ] oxy ] -5,6-dihydroxy-2- (4-hydroxy-3-methoxyphenyl) -4H-1-benzopyran-4-one&& |
|CAS=151750-86-6 | |CAS=151750-86-6 | ||
|KNApSAcK=C00004517 | |KNApSAcK=C00004517 | ||
}} | }} | ||
Latest revision as of 09:00, 22 September 2008
| Flavonoid Top | Molecule Index | Author Index | Journals | Structure Search | Food | New Input |
Upper classes : FL Flavonoid : FL3 Flavone : FL3FEC 6-Hydroxyluteolin and O-methyl derivatives (85 pages) : FL3FECGS O-Glycoside (52 pages)
| IDs and Links | |
|---|---|
| LipidBank | [1] |
| LipidMaps | [2] |
| CAS | 151750-86-6 |
| KEGG | {{{KEGG}}} |
| KNApSAcK | |
| CDX file | |
| MOL file | FL3FECGS0041.mol |
| 6-Hydroxyluteolin 3'-methyl ether 7- [ 6"- (3-hydroxy-3-methylglutaryl) glucoside ] | |
|---|---|
| |
| Structural Information | |
| Systematic Name | 7- [ [ 6-O- (4-Carboxy-3-hydroxy-3-methyl-1-oxobutyl) -beta-D-glucopyranosyl ] oxy ] -5,6-dihydroxy-2- (4-hydroxy-3-methoxyphenyl) -4H-1-benzopyran-4-one |
| Common Name |
|
| Symbol | |
| Formula | C28H30O16 |
| Exact Mass | 622.153384912 |
| Average Mass | 622.5282 |
| SMILES | COc(c(O)4)cc(cc4)C(O1)=CC(c(c2O)c1cc(O[C@H](O3)[C@ |
| Physicochemical Information | |
| Melting Point | |
| Boiling Point | |
| Density | |
| Optical Rotation | |
| Reflactive Index | |
| Solubility | |
| Spectral Information | |
| Mass Spectra | |
| UV Spectra | |
| IR Spectra | |
| NMR Spectra | |
| Chromatograms | |
Species Information
| Species-Flavonoid Relationship Reported | ||||||||
|---|---|---|---|---|---|---|---|---|
|